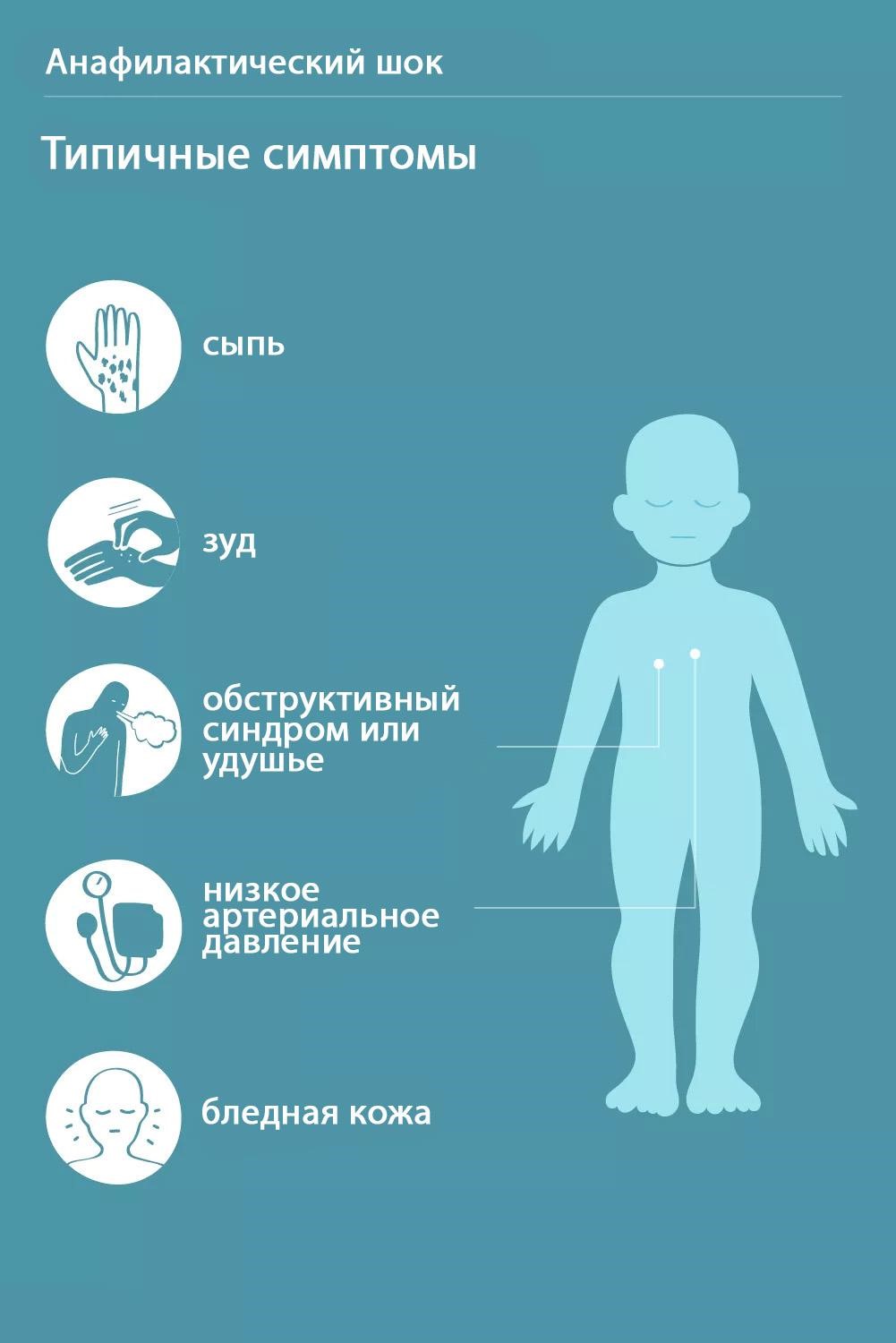
SergDoc | 0 | 4,336 | |||
: luidhen | 2 | 8,302 | |||
Tusia | 1 | 23,811 | |||
SergDoc | 0 | 10,390 | |||
SergDoc | 0 | 12,188 | |||
SergDoc | 34 | 202,797 | |||
jjull | 17.11.2016 13:15 jjull | 37 | 30,919 | ||
: Aspartam | 8 | 12,269 | |||
: luidhen | 3 | 46,587 | |||
luidhen | 22.08.2012 00:38 | 5 | 22,578 | ||
: . | 1 | 56,260 | |||
AlexGold | 1 | 45,157 | |||
: | 15.07.2011 13:18 | 1 | 22,475 | ||
skeptic | 64 | 49,062 | |||
alenkiy | 20.10.2009 12:09 . | 143 | 129,845 | ||
29.04.2009 01:11 | 0 | 23,435 | |||
nagisheva | 1 | 310 | |||
OlgaNLN | 2 | 372 | |||
Arabesque20 | 3 | 473 | |||
naaatka | 7 | 570 | |||
Vikolya | 2 | 313 | |||
Irinka1983 | 4 | 444 | |||
MariNochKa_N | 1 | 179 | |||
MariNochKa_N | 4 | 341 | |||
jabr | 1 | 248 | |||
Frosjka | 1 | 317 | |||
VestaNL | 1 | 425 | |||
Dubrowa | 8 | 1,516 | |||
Vita34 | 1 | 488 | |||
MirzoevaMasha | 1 | 332 | |||
Marimih | 9 | 774 | |||
MasterPeace | 10 | 1,323 | |||
Oliamusik | 13 | 830 | |||
Vikitinka | 3 | 530 | |||
KattyA | 1 | 494 | |||
dolka_22 | 3 | 460 | |||
nagisheva | 1 | 249 | |||
OlegV | 1 | 403 | |||
Natalimimoza | 1 | 341 | |||
Gulnazik | 1 | 298 | |||
Alena37 | 5 | 428 |
(https://forums.rusmedserv.com/index.php)
— — A/h2N1/2009 (https://forums.rusmedserv.com/forumdisplay.php?f=179)
— — ( ) (https://forums.rusmedserv.com/showthread.php?t=111921)
( )
!
13 .
: ( ), 75 ( ).
— «» — , .
?
.
| LupusDoc | 23.10.2009 14:08 |
.
| Carolain | 24.10.2009 16:07 |
, ,
. , . , , . .
, :
» , . . , , , , , . , , . , — , . , . — . , — . , .»
, , , .
2 , . , — . — .
obstetric AND flu vaccination AND safety , 1 , -, , .
( ), , , .
— . .
.
— KOL , — 
| LupusDoc | 24.10.2009 18:15 |
:
Carolain ( 890094)
, :
» , . . , , , , , . , , . , — , . , . — . , — . , .»
, . . , , CDC [ . ]
:
:
Pregnant Women and Neonates
Pregnant women have protective levels of anti-influenza antibodies after vaccination (147,148). Passive transfer of anti-influenza antibodies that might provide protection from vaccinated women to neonates has been reported (147,149—151). A retrospective, clinic-based study conducted during 1998—2003 documented a nonificant trend toward fewer episodes of MAARI during one influenza season among vaccinated pregnant women compared with unvaccinated pregnant women and substantially fewer episodes of MAARI during the peak influenza season (148). However, a retrospective study conducted during 1997—2002 that used clinical records data did not indicate a reduction in ILI among vaccinated pregnant women or their infants (152). In another study conducted during 1995—2001, medical visits for respiratory illness among the infants were not substantially reduced (153). One randomized controlled trial conducted in Bangladesh that provided vaccination to pregnant women during the third trimester demonstrated a 29% reduction in respiratory illness with fever and a 36% reduction in respiratory illness with fever among their infants during the first 6 months after birth. In addition, infants born to vaccinated women had a 63% reduction in laboratory-confirmed influenza illness during the first 6 months of life (154). All women in this trial breastfed their infants (mean duration: 14 weeks).
— . , , . .
:
:
Pregnant Women and Neonates
FDA has classified TIV as a «Pregnancy C» medication, indicating that adequate animal reproduction studies have not been conducted.. Available data indicate that influenza vaccine does not cause fetal harm when administered to a pregnant woman or affect reproductive capacity. One study of approximately 2,000 pregnant women who received TIV during pregnancy demonstrated no adverse fetal effects and no adverse effects during infancy or early childhood (190). A matched case-control study of 252 pregnant women who received TIV within the 6 months before delivery determined no adverse events after vaccination among pregnant women and no difference in pregnancy outcomes compared with 826 pregnant women who were not vaccinated (148). During 2000—2003, an estimated 2 million pregnant women were vaccinated, and only 20 adverse events among women who received TIV were reported to VAERS during this , including nine injection-site reactions and eight systemic reactions (e.g., fever, ache, and myalgias). In addition, three miscarriages were reported, but these were not known to be causally to vaccination (191). Similar results have been reported in certain smaller studies (147,149,192), and a recent international review of data on the safety of TIV concluded that no evidence exists to suggest harm to the fetus (193). The rate of adverse events associated with TIV was similar to the rate of adverse events among pregnant women who received pneumococcal polysaccharide vaccine in one small randomized controlled trial in Bangladesh, and no severe adverse events were reported in any study group (154).
— ,
. 2 20 , 9 — 8 (, ). ?
— .
| Carolain | 24.10.2009 18:30 |
., , ? .
LupusDoc — , » , — , .» . — . , ( — ) . , .
| zaremadag | 24.10.2009 19:44 |
. : :ac:
| LupusDoc | 24.10.2009 21:22 |
:
Carolain ( 890270)
LupusDoc — , » , — , .» . — .
. , , ( …). . ( ) .
| Carolain | 24.10.2009 21:50 |
LupusDoc . . , :
( !!!) . , — » «. ? 1953 .
— ?
— , , , . , , , , .
— . , . .
— . — .
— ?
— . . , , . , «» , , «», . . . . , «» , . » » «PUBMED» ([ . ]). (original antigenic sin influenza) , . !!! , .
— » » [ . ]
.
| LupusDoc | 24.10.2009 22:35 |
, . .
— Nature [ . ] ( ) , » » .
| Carolain | 25.10.2009 11:52 |
, . , .
:
LupusDoc ( 890586)
, . .
— Nature [ . ] ( ) , » » .
, . . — , . , , , , .
. , , . .
— . . . — , . .
vBulletin® 3.
Copyright ©2000 — 2021, Jelsoft Enterprises Ltd.
SergDoc | 84 | 113,244 | |||
SergDoc | 3 | 24,307 | |||
: AlexGold | 3 | 57,162 | |||
blacknataly | 8 | 54,632 | |||
luidhen | 0 | 15,689 | |||
luidhen | 0 | 17,824 | |||
luidhen | 0 | 11,026 | |||
skeptic | 23.05.2011 03:43 | 53 | 78,324 | ||
skeptic | 08.12.2010 19:36 easl | 65 | 82,293 | ||
: AlexGold | 08.04.2010 09:09 . | 9 | 71,440 | ||
: skeptic | 1 | 47,616 | |||
: Rodionov | 01.11.2009 05:01 | 10 | 100,270 | ||
: . | 8 | 52,947 | |||
: skeptic | 24.09.2009 10:49 Hard | 4 | 23,035 | ||
AlexGold | 2 | 41,328 | |||
AlexGold | 41 | 88,717 | |||
skeptic | 10.02.2009 20:21 | 8 | 48,169 | ||
jwassa | 14 | 64,268 | |||
bill | 05.08.2008 10:15 bill | 107 | 96,807 | ||
luidhen | 02.10.2019 10:27 easl | 2 | 4,885 | ||
luidhen | 1 | 12,744 | |||
luidhen | 0 | 4,041 | |||
SergDoc | 1 | 9,515 | |||
Elena_Melnik | 0 | 8,765 | |||
crocodile | 22.07.2014 21:47 | 3 | 10,381 | ||
Dkno | 2 | 16,601 | |||
AlexGold | 1 | 17,270 | |||
alcorstar | 6 | 12,144 | |||
BBC | 16 | 38,337 | |||
Valentine11 | 17 | 24,748 | |||
skeptic | 3 | 43,310 | |||
luidhen | 08.03.2012 20:54 | 14 | 23,271 | ||
skeptic | 3 | 25,946 | |||
skeptic | 14 | 25,559 | |||
skeptic | 79 | 92,261 | |||
skeptic | 18.12.2010 01:16 | 6 | 25,534 | ||
skeptic | 371 | 233,284 | |||
Kat | 1 | 18,905 | |||
skeptic | 13 | 22,621 | |||
skeptic | 18.11.2010 01:03 Hard | 1 | 9,817 | ||
skeptic | 0 | 14,160 | |||
skeptic | 0 | 10,584 | |||
Aspartam | 07.11.2010 19:18 | 11 | 13,676 | ||
skeptic | 3 | 10,401 |
- Мустафин Р. И., Буховец А. В., Протасова А. А., Шайхрамова Р. Н., Ситенков А. Ю., Семина И. И. Сравнительное исследование поликомплексных систем для гастроретентивной доставки метформина. Разработка и регистрация лекарственных средств. 2015; 1(10): 48–50.
- М.П. Киселева, З.С. Шпрах, Л.М. Борисова и др. Доклиническое изучение противоопухолевой активности производного N-гликозида индолокарбазола ЛХС-1208. Сообщение I // Российский биотерапевтический журнал. 2015. № 2. С. 71-77.
- М.П. Киселева, З.С. Шпрах, Л.М. Борисова и др. Доклиническое изучение противоопухолевой активности производного N-гликозида индолокарбазола ЛХС-1208. Сообщение II // Российский биотерапевтический журнал. 2015. № 3. С. 41-47.
- https://forums.rusmedserv.com/forumdisplay.php?f=172.
- https://forums.rusmedserv.com/printthread.php?t=111921.
- https://forums.rusmedserv.com/forumdisplay.php?f=131.
- Daremberg, «Histoire des sciences médicales» (П., 1966).
- Мустафин Р. И., Буховец А. В., Протасова А. А., Шайхрамова Р. Н., Ситенков А. Ю., Семина И. И. Сравнительное исследование поликомплексных систем для гастроретентивной доставки метформина. Разработка и регистрация лекарственных средств. 2015; 1(10): 48–50.